Hydrogen Atoms
Hydrogen Atoms
Since hydrogen cars are powered by hydrogen atoms, we thought it would be good to have a primer on hydrogen atoms. Many people have some concept of what hydrogen is either from schooling or independent research, but most don’t know that hydrogen (H1) is the simplest and most prevalent element in the universe.
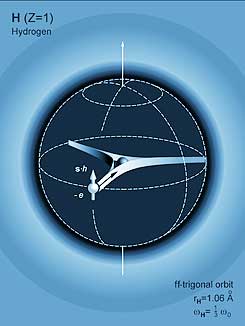 |
A hydrogen atom is composed of one proton surrounded by one electron. It doesn’t get any simpler than this!
|
Physics theorists say that hydrogen was first created shortly after the Big Bang. Covalent bonds are formed when hydrogen atoms join other atoms to create molecules such as water molecules (which is the waste dispensed from hydrogen cars).
There are many theories regarding hydrogen atoms, which we won’t get into here, such as the Coulomb Force, Schrodinger Equation, Bohr Model. Suffice it to say, investigating the physical properties of hydrogen atoms from the quantum physics model or Newtonian mechanics model or even particle physics theoryis quite an undertaking left for those deeply embedded in those fields.
For our purposes here, it is important to know how a hydrogen atom produces energy through a hydrogen fuel cell. A fuel cell generally consists of two electrodes sandwiched around an electrolyte. Hydrogen atoms pass over one electrode and oxygen atoms over the other generating electricity, water and heat. The hydrogen atom splits into a proton and electron when excited by a catalyst. The hydrogen protons and electrons take different paths to the cathode, which is where the oxygen molecule enters. The electrons create a separate current,which is used by the fuel cell before it returns to the cathode and is reunited with the proton and oxygen to form a water molecule.
So, yes, the hydrogen atom is split to create a small amount of energy, then quickly reunited to and merged with oxygen to create water. Since small amounts of energy are created by this process (as opposed to a hydrogen bomb where huge amounts of energy is created) the fuel cells are generally grouped in stacks in order to generate enough electricity to power a car.
|
Because hydrogen atoms are not found freely in nature in any significant amounts, they have to be separated from other compounds such as water or natural gas first, before being sent through a fuel cell. |
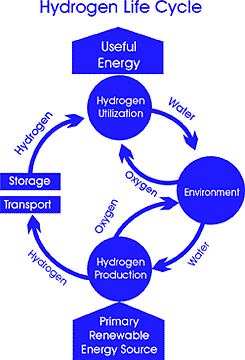 |
It is the process of generating enough hydrogen economically and distributing it that will be the greatest challenge in a hydrogen economy.
Written by Hydro Kevin Kantola
Leave a Reply
You must be logged in to post a comment.